Following two recent publications in the Journal of Bronchology and Interventional Pulmonology, one in July 2020 and another in October 2020, Body Vision has garnered significant interest from pulmonologists and key opinion leaders worldwide. A common question we receive is, “How is your technology different from Electromagnetic Navigation Bronchoscopy (ENB), and what are the benefits of using Body Vision in my practice?”
These inquiries are crucial and have been central to our mission from the start. Body Vision is more than just a navigation system; it is an advanced, real-time, intraoperative imaging modality. Unlike existing technologies that depend on a pre-operative CT scan to create a virtual navigation target and, if successful, use a “cloud biopsy” approach, Body Vision transforms any C-arm into a real-time intraoperative CT imaging system. This allows for direct visualization of the actual lesion during the procedure, eliminating CT-to-body divergence and enabling visual confirmation of tool-in-lesion before biopsy. This innovative approach is revolutionizing diagnostic bronchoscopy by providing evidence-based biopsies using equipment already present in most bronchoscopy suites.
The primary goal of diagnostic bronchoscopy is to obtain an adequate tissue sample from the suspicious lesion so a pathologist can accurately determine if there is malignancy. Until our advanced imaging technology, no readily accessible solution allowed bronchoscopists to confidently reach the pulmonary lesion, confirm tool placement within the lesion before biopsy, and provide the real-time visualization necessary for an evidence-based biopsy approach.
Introduction: Definitions and Etiology
Reliable diagnostic results from a navigational bronchoscopy procedure are crucial for enhancing patient care. A definitive diagnosis achieved through diagnostic bronchoscopy can lead to earlier treatment and significantly improve survival rates. Additionally, obtaining a definitive diagnosis enhances the patient experience by reducing the need for further, potentially more invasive procedures and providing the certainty that lung cancer patients seek.
Historically, there have been two key criteria for evaluating diagnostic results:
Diagnostic Yield - This can vary depending on the specificity of what is considered "diagnostic." Essentially, it is defined as the percentage of procedures that provide a definitive explanation for the presence of a pulmonary lesion, whether benign or malignant.
Localization - This refers to the ability to accurately navigate to a point in 3D space, whether it is a real or virtual target. Like diagnostic yield, it is typically expressed as a percentage (e.g., the percentage of procedures in which the bronchoscopist successfully reached the target).
Currently, high localization does not necessarily correlate with high diagnostic yield due to the dynamic nature of the lung and the reliance on preoperative CT scans to establish a virtual target. This can result in bronchoscopists navigating to the virtual target but not the actual lesion, leading to biopsy samples that do not provide a definitive diagnosis.
Only through real-time, intraoperative imaging that allows clinicians to visualize the actual lesion, its location, and the position of their tools relative to the lesion can bronchoscopists maximise the likelihood of a definitive diagnosis and achieve a high diagnostic yield.
Computed tomography (CT)-guided transthoracic needle aspiration (TTNA) has demonstrated a diagnostic yield of up to 92.1%* because interventional radiologists performing the procedure use CT to visualize both the lesion and the aspiration needle, ensuring the needle is in the lesion before taking a tissue sample. The lack of similar intraprocedural imaging in diagnostic bronchoscopy is one reason the procedure has only achieved a comparatively poor diagnostic yield of 72%, even with electromagnetic navigation. ³
However, CT-guided TTNA is associated with higher rates of complications, such as pneumothorax. Therefore, an approach combining the low patient risk of traditional navigational bronchoscopy with the high diagnostic rates and consistency of TTNA would be ideal.
In 2014, Body Vision Medical was founded to address the unmet clinical need for early, definitive lung cancer diagnosis. Body Vision's AI-driven intraoperative imaging gives bronchoscopists the real-time intraoperative imaging they previously could not get with a traditional C-arm, combining the low risk of navigational bronchoscopy with the clinical outcomes of TTNA.
Since its launch, Body Vision has transformed bronchoscopy methods by making it possible to see the lesion and its exact location, removing the need for CT-to-body divergence, and offering real-time tool-in-lesion confirmation, which is essential for ensuring that tissue samples are taken from within the lesion and increasing the chance of a definitive diagnosis.
In this blog, we will explore Body Vision technology in depth and how this advanced technology enables clinicians to maximize diagnostic yield. First, it is essential to understand key terms and definitions related to this process.
Bronchoscopy - A procedure allowing doctors to examine a patient's lungs and airways. It involves inserting a thin tube through the nose or mouth, leading to the throat and lungs. Bronchoscopy is typically performed by a specialist to identify lung disorders or cancer, and to locate abnormal areas in the airways that need further examination.
Pulmonary Lesion - An abnormal growth in the lung, that can be benign (noncancerous) or malignant (cancerous). Common causes of benign nodules include granulomas (inflamed tissue clumps) and hamartomas (benign lung tumors). Malignant nodules are often caused by lung cancer or metastatic cancer. Accurate testing, usually through tissue sampling, is required to determine whether a lesion is benign or malignant.
Fluoroscopy - An imaging technique using X-rays to create continuous, real-time images of the inside of the body, similar to an X-ray movie.
Computed Tomography (CT) - A CT scan combines X-ray images taken from various angles around the body and uses computer processing to create cross-sectional images of bones, blood vessels, and soft tissues. CT scans provide more detailed information than plain X-rays and are used in various diagnostic tests to detect diseases, cancers, and more.
Transthoracic Needle Aspiration (TTNA) - An image-guided biopsy technique used to diagnose thoracic diseases. TTNA employs CT or ultrasound for real-time image guidance as a biopsy needle is introduced through the chest wall to take tissue samples from a suspicious lesion. Historically, TTNA has a high diagnostic rate of up to 90% but also a high complication rate of pneumothorax (collapsed lung) at nearly 25%.²
C-Arm Based Tomography™ Technology - Body Vision's CABT™ technology uses a proprietary AI-driven computer imaging algorithm to transform 2D X-ray images from any C-arm into 3D intraoperative CT scans. This technology provides real-time visualization of the lesion and its location during the procedure, enabling accurate navigation and visual confirmation of tool-in-lesion before and during biopsy.
The Development of Body Vision’s Tool-in-Lesion Technology
Body Vision emerged from the need for imaging technology that enables bronchoscopists to achieve diagnostic yields comparable to interventional radiologists while addressing the quadruple aim of healthcare—delivering superior clinical outcomes and patient experience at a lower cost using existing medical staff. Before Body Vision, innovations in pulmonology focused mainly on navigation, with electromagnetic navigation bronchoscopy (ENB) being a leading technology. Although ENB significantly improved the accuracy of navigating to a virtual target, it relied on a pre-operative CT scan to set this target, making it vulnerable to CT-to-body divergence—the discrepancy between the pre-operative CT and the actual lesion location during bronchoscopy. To resolve this issue, Body Vision combined intraoperative CT imaging with augmented fluoroscopy.
Fluoroscopy was an excellent choice because it is widely used in biopsy procedures and utilizes a common X-ray device called a C-arm. It continuously takes images of the body using X-rays, providing true real-time imaging. However, pulmonary lesions are typically hard to see or entirely invisible under fluoroscopy. Theoretically, since the C-arm is an X-ray device like a CT scanner, a computer algorithm could potentially fuse 2D X-ray images from different angles into an accurate 3D scan similar to a CT scanner. Nevertheless, the C-arm is less precise than the CT scanner, and CT scanners vary by model and manufacturer, each with different configurations.
To overcome these challenges, Body Vision developed an AI-driven computer imaging algorithm. As the AI system processed more clinical imaging data, it learned to recognize lesion borders, bronchoscopic tools, anatomical landmarks, and other key aspects to enhance the image quality derived from a C-arm. This technology for tomographically visualizing radiolucent lesions with a C-arm was named C-Arm Based Tomography (CABT™). Since the innovation was software-based, Body Vision’s solution is hardware-agnostic and designed to integrate easily into procedural rooms and interface with existing bronchoscopic equipment.
The Body Vision System
The Body Vision system was designed not only to ensure navigational accuracy and maximize diagnostic yield by enabling visual confirmation of tool-in-lesion during biopsy but also to seamlessly integrate into procedure rooms. Recognizing that procedure rooms are often crowded with hardware and electronic equipment, the Body Vision system has a small footprint and interfaces with existing bronchoscopy platforms, C-arms, and other equipment without needing additional equipment beyond the Body Vision itself.
The LungVision™ System Components:
Main Unit: The core of the LungVision™ system, performing the image processing needed to transform a traditional 2D C-arm into a real-time, intraoperative CT imaging system. Its small footprint ensures it remains unobtrusive in the bronchoscopy suite.
Tablet: The LungVision™ tablet allows clinicians to wirelessly control the system and manage the procedure from anywhere in the room.
Positioning Board: A board with three layers of radiopaque beads placed under the patient during the procedure. This enables the LungVision™ system to identify the 3D spatial arrangement with a single fluoroscopy image. As the system is entirely imaging-based, the board contains no sensors or electronics.
Disposable Procedural Kit: For those using Body Vision as a complete diagnostic bronchoscopy platform, the disposable procedural kit offers an extended working channel for greater maneuverability, visibility of the catheter under fluoroscopy, and access to the lung periphery with a traditional manual catheter setup. The absence of sensors or cameras maximizes flexibility, avoids interference with metal, and makes the procedural kit more economical.
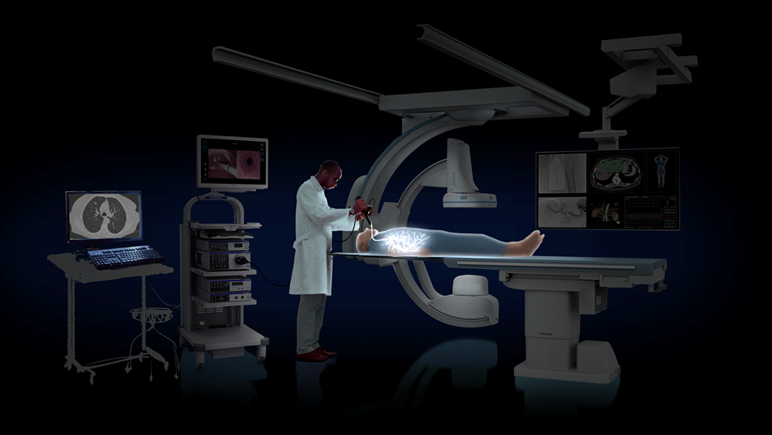
The Procedure
A biopsy is usually needed for a definitive diagnosis if a suspicious pulmonary lesion is identified during screening. Diagnostic bronchoscopies have minimal invasiveness and low complication rate, making it an increasingly preferred method. The purpose of Body Vision's LungVisionTM system is to acquire sufficient tissue so that pathology can determine with certainty whether the lung patient has cancer. The Body Vision procedure consists of five steps:
- Pre-Procedure Planning
- Registration
- Navigation
- Tool-in-Lesion Confirmation
- Image-Guided Biopsy
1) Pre-Procedure Planning
A pre-operative CT is uploaded into the Body Vision system. In order to provide guidance for iso-centering the c-arm over the lesion during the procedure, the system uses the pre-operative CT scan to determine the airways. It then uses the physician's marking of the lesion and its boundaries to approximate the lesion's location before the procedure.
Preoperative CT is uploaded
The lesion is identified on the preoperative scan, and the lesion and lesion boundaries are marked.
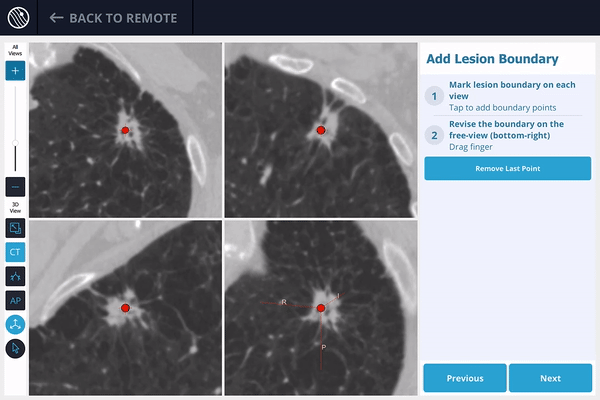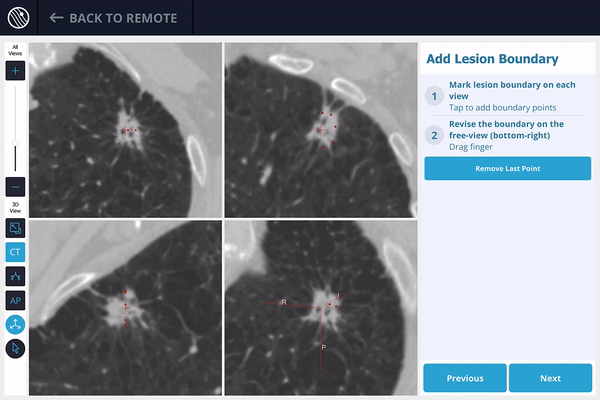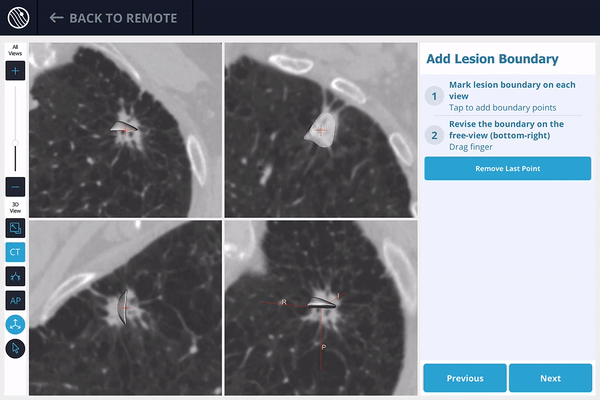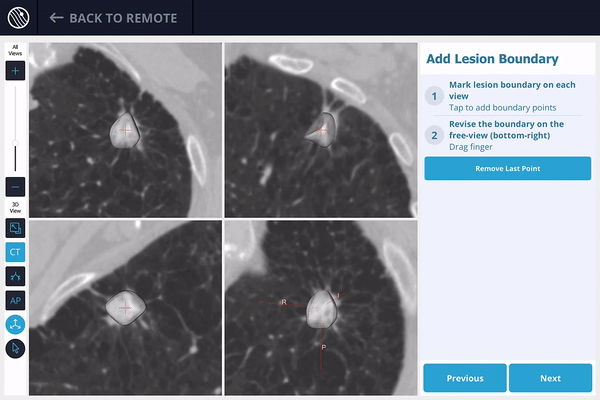
The LungVision™ System derives airway information from the pre-operative CT and displays the bronchial tree such that the clinician can choose the preferred pathway, with LungVision™ highlighting the pathway from the entrance of the trachea to the lesion.
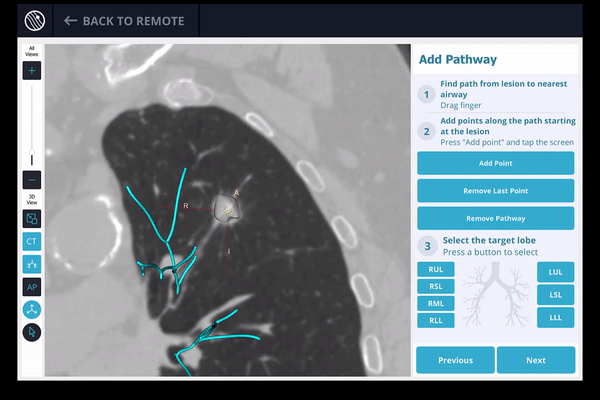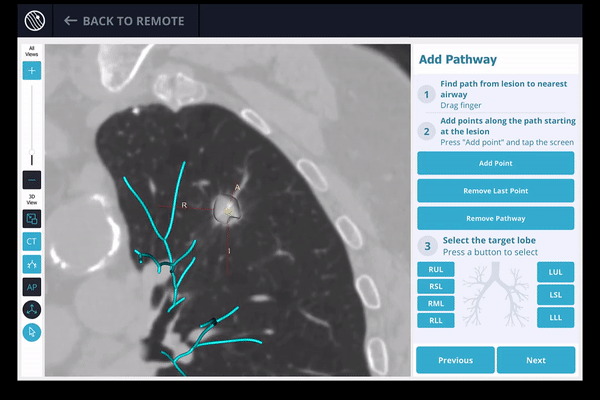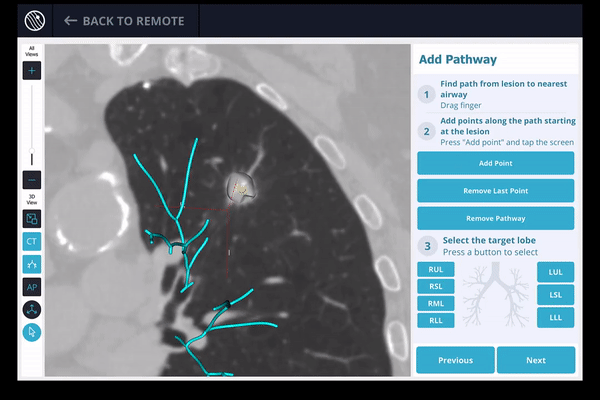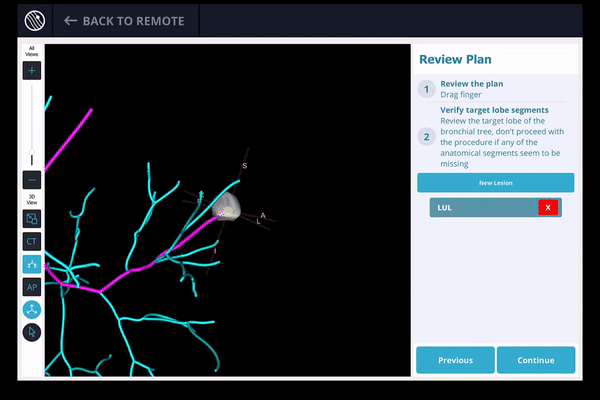
Once the pre-procedure planning is complete, a virtual bronchoscopy simulation is provided as a summary of the preferred pathway to the lesion.
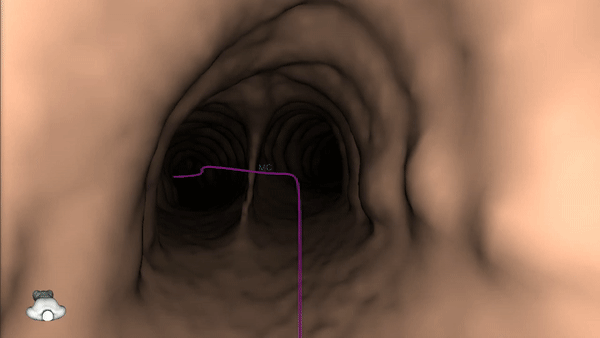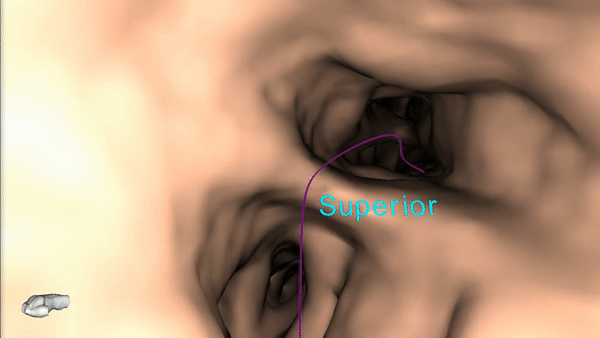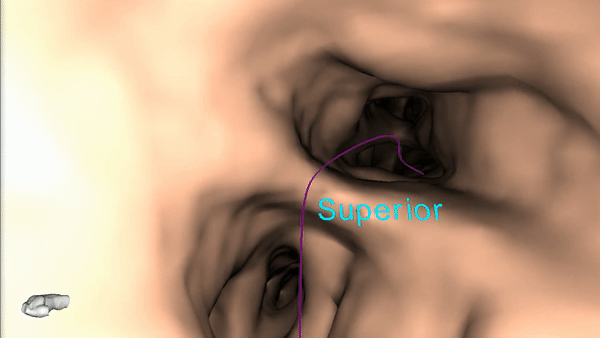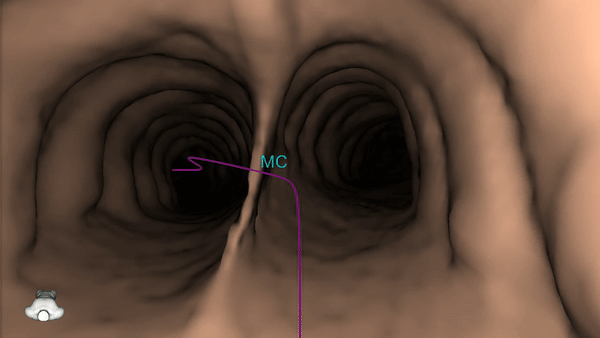
The procedure is ready to begin.
2) Registration
Body Vision's C-arm-based computational tomography (CABT) technology provides intraoperative 3D images using any conventional C-arm. Registration of the Body Vision system involves performing a CABT scan of the main carina, followed by a second CABT scan centered around the lesion. This process allows for the visualization of the actual lesion and its location, including radiolucent lesions that are typically not visible under fluoroscopy, before navigation begins.
3) Navigation
Augmented fluoroscopy displays the actual lesion and the pre-planned pathway as an overlay. The virtual bronchoscopy simulation from the planning step can be accessed at any time during the procedure to provide a bronchoscopic view of the preferred path to the lesion. When the bronchoscope reaches the point where its diameter is larger than the airways leading to the lesion, the disposable LungVision™ Procedural Kit is used to extend the working channel, providing the maneuverability needed to reach the lesion. Under real-time, augmented fluoroscopy, which highlights the lesion location, preferred pathway, and adjunctive airways, the clinician guides the distal tip of the LungVision™ sheath to the lesion. Radiopaque markers on the LungVision™ sheath make it easy to identify the sheath and its tip under fluoroscopy, aiding in navigation.
4) Tool-in-Lesion Confirmation
Once the LungVision™ sheath reaches the lesion location, a biopsy tool is inserted. Before sampling, it’s recommended to confirm that the biopsy tool is visually inside the lesion in multiple 3D planes using Body Vision’s CABT™ technology. This technology allows users to see the spatial relationship between the lesion and the biopsy tool tip. The interactive 3D View functionality presents this data as a 3D reconstruction, aiding those who struggle with interpreting 3D scans in three separate orthogonal planes to determine if tool-in-lesion has been achieved. If not, adjustments can be made to the bronchoscope and/or sheath. This approach eliminates concerns about accuracy and localization since the clinician can visualize the actual lesion, avoiding CT-to-Body divergence by enabling intraoperative visualization with a conventional C-Arm.
LungVision™ technology allows physicians to use any instrument of their choice with its high compatiblility. Physicians can also extend a Radial EBUS probe (r-EBUS) down the LungVision™ sheath before inserting the biopsy tool for additional lesion location confirmation If desired
5) Image-Guided Biopsy
Once the biopsy tool tip is confirmed inside the lesion in multiple 3D planes, the biopsy is performed under augmented fluoroscopy, allowing the clinician to verify in real-time that samples are taken from within the lesion. With LungVision™, the biopsy process is image-guided and evidence-based. There is no longer a need to systematically sample from a wide area around a virtual target in hopes of obtaining adequate tissue samples, as LungVision enables visualization of the actual lesion. The LungVision™ procedural kit is compatible with any off-the-shelf biopsy tools with an outer diameter of 1.9mm or less.
The Advantages of AI-Powered, Intraoperative CT Imaging
Body Vision’s AI-powered, intraoperative CT imaging technology is designed to enhance diagnostic accuracy and improve clinical outcomes, providing lung patients with the certainty they need and enabling timely treatment if necessary. Here are some key benefits of using this advanced technology:
Early Lung Cancer Treatment - Lung cancer is the second most common cancer in the U.S., accounting for 25% of all cancer deaths annually, more than colon, breast, and prostate cancers combined. With 70% of lung cancers detected at later stages, the 5-year survival rate remains at 20% despite advancements in medical technology, smoking cessation efforts, and lung cancer screening. Increasing lung cancer screening in high-risk populations, coupled with the ability to definitively diagnose patients earlier, can allow more patients to receive timely treatment, thereby improving long-term survival rates.
Seamless Integration - The Body Vision system is designed for easy integration into existing procedural rooms, bronchoscopy setups, and workflows
Utilization of Artificial Intelligence - Body Vision Medical has pioneered the use of AI in navigation bronchoscopy. By harnessing AI, Body Vision has developed a cost-effective solution that enables medical teams to deliver superior clinical outcomes and patient experiences. Unlike hardware-intensive solutions such as robotics and cone-beam CT, Body Vision’s software-based approach not only reduces costs but also allows for rapid feature and function iterations that directly impact user experience and clinical results.
Advantages of Body Vision's Tool-In-Lesion Technology | Case Study
Body Vision’s LungVision™ AI-powered, intraoperative CT imaging system stands out due to its ability to maximize diagnostic yield while leveraging existing equipment, making the solution cost-effective. For years, navigating to and confidently biopsying peripheral lung lesions was challenging because bronchoscopists could not see the lesion or confirm tool-in-lesion prior to biopsy.
Body Vision addresses this challenge by allowing physicians to visualize peripheral pulmonary lesions in real-time during procedures, eliminating CT-to-body divergence and providing visual confirmation of tool-in-lesion before biopsy. Numerous clinical studies and peer-reviewed literature support the concept that real-time, intraoperative 3D imaging is crucial for achieving higher diagnostic yields, regardless of the navigation modality.
Here are some of the key findings from the case study:
Lesion Characteristics:
- Lesion Size (diameter): 12 mm
- Lesion Location: Right upper lobe (RUL) lesion
- Bronchus Sign: No
- Visible on Fluoro: No
- REBUS Verification: Yes, after Body Vision enabled navigation to the lesion.
Case Information
- Full Procedure Time: 45 minutes
- ROSE: Sampling was performed and was positive for non-small cell lung cancer (NSCLC)
- Final Pathology Report: Final pathology was adenocarcinoma of the lung.
A 69-year-old patient, a current smoker with chronic obstructive pulmonary disease (COPD), underwent routine CT screening, revealing a suspicious nodule in the right upper lobe. Further positron emission tomography (PET) scanning confirmed hyper-metabolic activity in the area. Consequently, the patient was scheduled for a robotic-assisted navigation bronchoscopy using the Auris Monarch platform and Body Vision system for a biopsy of the peripheral lung nodule. Here is a detailed look at the procedure:
Planning - Initially, a preoperative CT scan was uploaded into the Body Vision imaging system, which is powered by AI. The lesion was then marked and contoured to estimate its shape, size, and location accurately.
Registration - After positioning the patient, registration was performed using two C-arm spins: one centered around the main carina and the other iso-centered on the lesion. This resulted in a CABT™ scan, confirming the lesion's presence and location before navigation.
Navigation - The Auris Monarch bronchoscopy platform was navigated to the virtual target. A radial probe endobronchial ultrasound (r-EBUS) was advanced down the Monarch bronchoscope to locate the virtual target. However, this method did not detect the lesion at the target location, prompting the use of a C-arm and a CABT™ scan with the Body Vision system. This approach accurately determined the true lesion location, eliminating any CT-to-body divergence due to the real-time imaging capabilities of the Body Vision system.
r-EBUS Confirmation - The r-EBUS probe was re-inserted through the Monarch bronchoscope and extended to the lesion location identified by Body Vision. The probe's return confirmed the location determined by Body Vision.
Tool-In-Lesion Confirmation - The r-EBUS probe was withdrawn and replaced with biopsy forceps, which were extended into the pulmonary lesion. Body Vision and r-EBUS confirmed the forceps' position. A subsequent C-arm spin provided visual confirmation of the tool-in-lesion in multiple 3D planes, enabling the biopsy to proceed.
Biopsy - The biopsy was performed under real-time imaging using Body Vision’s augmented fluoroscopy, ensuring tissue samples were taken from within the lesion. On-site evaluation indicated the samples were consistent with non-small cell lung cancer, later confirmed by cytology and final pathology reports.
This case highlights how Body Vision’s AI-driven imaging seamlessly integrates into traditional diagnostic bronchoscopy workflows. Although robotic-assisted technology added stability to navigation, real-time 3D imaging was crucial for making necessary adjustments. In this case, a correction was required for the Monarch system.
By providing visualization of the lesion and its precise location, Body Vision enabled accurate repositioning of the bronchoscope, facilitating tool-in-lesion confirmation. This maximized the likelihood of obtaining tissue samples from within the lesion, leading to a definitive lung cancer diagnosis. With accurate results, the medical team could proceed with appropriate treatment and care. This case exemplifies how Body Vision's technology transforms diagnostic bronchoscopy, ensuring accuracy from the first attempt.
Conclusion
A novel approach was clearly needed to enhance the diagnostic yield of navigational bronchoscopy. Despite innovations in pulmonology, such as electromagnetic navigation, robotic bronchoscopy platforms, and cone-beam CT imaging, these solutions either partially address the issue or are too costly, complex, or limited in access for most bronchoscopists. Body Vision aimed to save lives by democratizing advanced medical technology, leading to the development of the LungVision™ AI-driven, intraoperative CT imaging platform. This platform meets the critical need for early and definitive lung cancer diagnosis and aims to facilitate effective, minimally-invasive treatment of lung lesions in the future.
The results are promising, but there is still room for improvement in user experience and customer training. Our goal is to enable users to achieve autonomy more quickly, allowing them to focus on delivering the best patient care. Additionally, we aim to enhance imaging quality. One significant advantage of our software-driven technology is the ability to implement continuous improvements, ensuring ongoing advancements in both functionality and user experience.
References
American Lung Association. (n.d.). What to Expect from a Lung Cancer Screening | Saved By The Scan. American Lung Association. Retrieved July 29, 2024, from https://www.lung.org/lung-health-diseases/lung-disease-lookup/lung-cancer/screening-resources/what-to-expect-from-lung-cancer-screening
DiBardino DM, Yarmus LB, Semaan RW. Transthoracic needle biopsy of the lung. J Thorac Dis. 2015 Dec;7(Suppl 4):S304-16.
Mehta AC, Hood KL, Schwarz Y, Solomon SB. The Evolutional History of Electromagnetic Navigation Bronchoscopy: State of the Art. Chest. 2018 Oct;154(4):935-947.
Source: https://bodyvisionmedical.com/articles/inside-body-vision-process






